The Gila Monster and the Billion-Dollar Molecule
How a desert lizard’s venom, ignored by every major pharmaceutical company for nearly a decade, became the template for the world’s most-prescribed weight-loss drugs.
[Note: Apologies we had a lapse in new articles due to various life issues. Back on track.]
In 1992, a biochemist named John Eng at the Veterans Administration Medical Center in the Bronx was running a chemical screen through Gila monster venom. He was not looking for a diabetes drug. He was running a systematic search for histidine-terminal peptides in animal secretions — the kind of methodical work that rarely makes headlines. What he found was a 39-amino acid molecule he named exendin-4, secreted from glands along the lower jaw of Heloderma suspectum, the only venomous lizard native to the United States.1
Eng’s paper, published in the Journal of Biological Chemistry, noted something strange: exendin-4 shared 53% of its amino acid sequence with a human gut hormone called glucagon-like peptide-1 (GLP-1), which had been characterized just a few years earlier.2 It appeared to bind the same receptor. The paper closed with a careful prediction: there likely existed “an endogenous mammalian analog to the exendin peptides.”
He was describing GLP-1 without knowing it had already been found.
The 2-Minute Problem
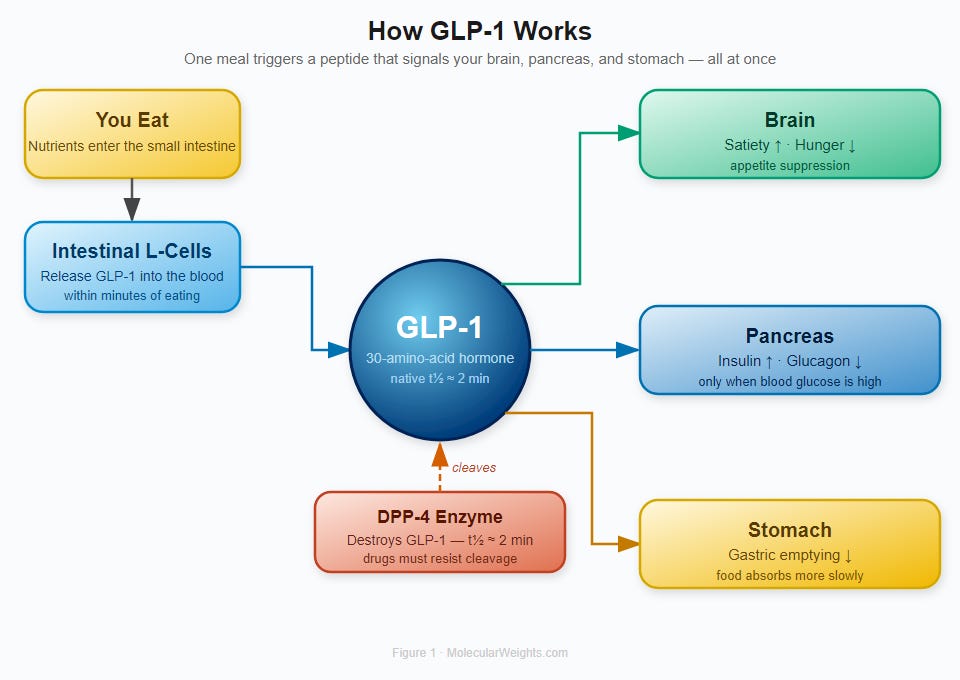
GLP-1 is a 30-amino acid incretin hormone, made by L-cells lining the distal small intestine.3 Within minutes of eating, nutrients arriving in the gut trigger a burst of GLP-1 release. The peptide spreads into the circulation and does five things nearly simultaneously: it drives insulin secretion from pancreatic beta cells, suppresses glucagon from alpha cells, slows gastric emptying, and sends satiety signals to the hypothalamus and brainstem, telling the brain the meal is enough.4 This coordination accounts for roughly 50–70% of the insulin response to an oral meal in healthy people, a phenomenon called the incretin effect.5
The problem is that GLP-1 lasts about 2 minutes in the bloodstream.6
An enzyme called DPP-4 circulates in the blood and cleaves the peptide almost immediately after it is released, cutting between the second and third amino acids and rendering it biologically inactive. Native GLP-1 was recognized as a potential drug target by the late 1980s. But infusing a molecule that disappears in 2 minutes is not practical medicine.
Figure 1. GLP-1 Signaling Cascade. After a meal, nutrients in the small intestine trigger L-cells to release GLP-1 into the bloodstream. The hormone acts simultaneously on the brain (satiety), pancreas (insulin release, glucagon suppression), and stomach (slowed emptying). DPP-4 enzyme cleaves GLP-1 with a half-life of approximately 2 minutes, which is why early drug development focused on DPP-4 inhibitors and on engineering longer-lived GLP-1 analogs.
The Lizard’s Advantage
Exendin-4 has one small but critical difference from human GLP-1: its second amino acid is glycine rather than alanine. That substitution blocks DPP-4 from cleaving it. Where human GLP-1 disappears in 2 minutes, exendin-4 persists for about 2.4 hours.7
In 1993, a German laboratory confirmed what Eng’s paper had implied: exendin-4 was not merely structurally similar to GLP-1 — it was a high-potency agonist at the human GLP-1 receptor, stimulating the same cAMP signaling cascade in insulin-secreting beta cells and amplifying glucose-induced insulin secretion.8 The Gila monster had been running the same pharmacological experiment on its own digestive biology, across evolutionary time, that pharmacologists were trying to replicate in the lab.
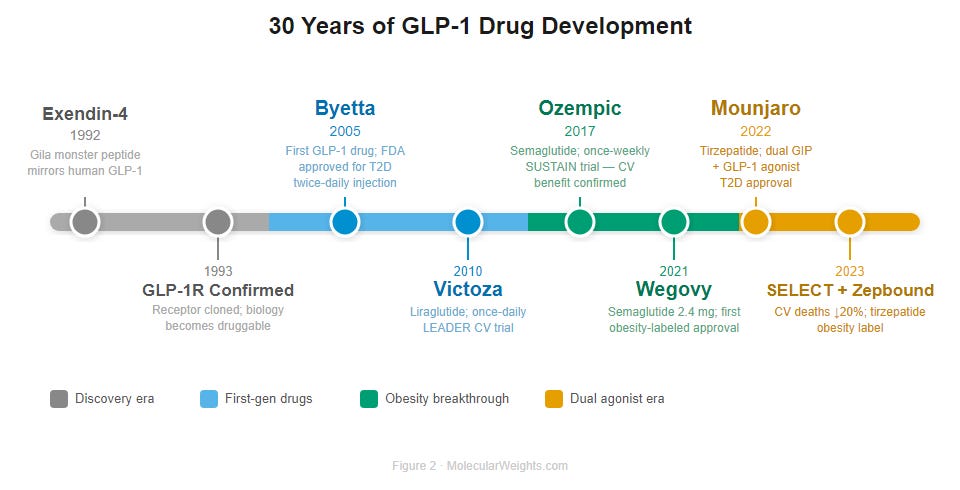
Pharmaceutical companies were largely uninterested. Peptide drugs in the early 1990s were considered impractical. Even exendin-4’s improved 2.4-hour half-life meant twice-daily injections at best. Eng’s work sat largely dormant for nearly a decade, until animal studies around 1999 showed the lizard peptide improved glucose tolerance, expanded beta cell mass, and reduced food intake in rodents.9
A synthetic version of exendin-4, called exenatide (brand name Byetta), became the first GLP-1 receptor agonist approved by the FDA on April 28, 2005.10
Engineering for a Longer Life
Twice-daily injection was never going to be a mass-market product. The next question was how to keep a peptide alive in the bloodstream long enough to work with less frequent dosing.
Figure 2. From Lizard Venom to Billion-Dollar Drug: 30 Years of GLP-1. Thirty-year arc from Gila monster venom to a global obesity drug class. Timeline is not to proportional scale. Colors denote development generation: gray = foundational pharmacology; blue = first-generation GLP-1 receptor agonists; green = semaglutide era; orange = dual GLP-1/GIP agonists and cardiovascular outcomes. Abbreviations: GLP-1 RA, glucagon-like peptide-1 receptor agonist; GIP, glucose-dependent insulinotropic polypeptide; MACE, major adverse cardiovascular events.
The answer came from Novo Nordisk, who engineered liraglutide (Victoza, FDA approved January 2010) by attaching a C16 fatty acid chain to a modified GLP-1 backbone. The chain binds serum albumin in the blood. Albumin is a large protein the kidneys don’t filter; drugs that hitchhike on it stay in circulation far longer. Liraglutide’s half-life extended to 13 hours — long enough for once-daily injection.11
Semaglutide, whose chemistry Novo Nordisk published in 2015 and which received FDA approval in December 2017 as Ozempic for type 2 diabetes, pushed the concept further. The team made three simultaneous changes to the GLP-1 backbone: a position-8 amino acid substitution to block DPP-4 cleavage, an arginine substitution to prevent fatty-chain attachment at the wrong lysine, and attachment of a C18 fatty diacid chain via a short PEG linker at lysine-26.12 The fatty diacid binds albumin far more tightly than liraglutide’s monoacid. The result: a plasma half-life of approximately 7 days.13 Once-weekly injection. Steady state in four weeks.
One molecule. Three modifications. Seven days.
What the Numbers Look Like
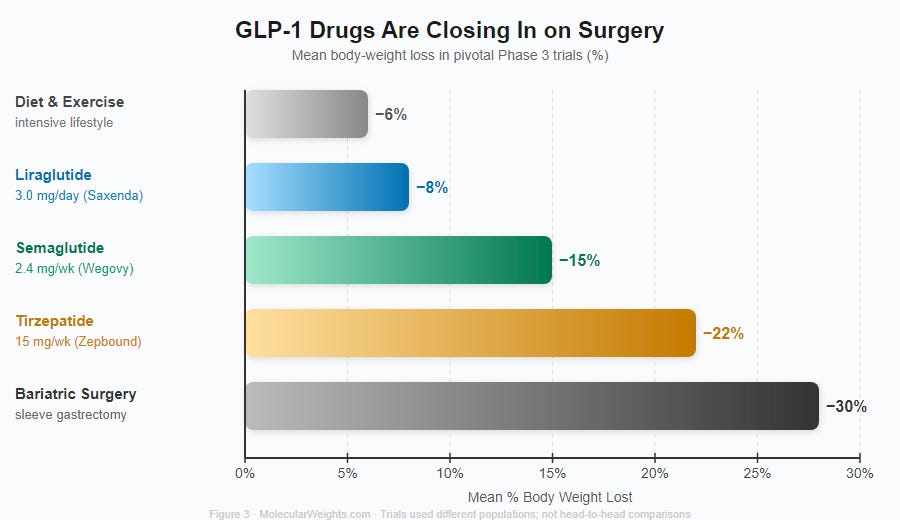
The STEP 1 clinical trial, published in the New England Journal of Medicine in 2021, enrolled 1,961 adults with obesity and no diabetes. After 68 weeks on semaglutide 2.4 mg weekly, participants lost an average of 14.9% of their body weight versus 2.4% on placebo. More than half the semaglutide group lost at least 15%.14 The two-year STEP 5 trial showed the losses were maintained: −15.2% at 104 weeks.15
Figure 3. GLP-1 Drugs Are Closing In on Surgery. Mean body-weight loss from each drug’s pivotal Phase 3 trial (SCALE for liraglutide 3.0 mg; STEP 1 for semaglutide 2.4 mg; SURMOUNT-1 for tirzepatide 15 mg). Bariatric sleeve gastrectomy shown as surgical benchmark. Trials used different populations and follow-up lengths; not head-to-head comparisons.
The cardiovascular data were even more striking. The SELECT trial, reported in the New England Journal of Medicine in 2023, followed 17,604 adults with obesity and pre-existing cardiovascular disease but no diabetes. Those on semaglutide 2.4 mg weekly had a 20% lower relative risk of major cardiovascular events, including heart attack and stroke, over a median follow-up of nearly 40 months.16 The effect was large enough to matter clinically, not just statistically. Readers familiar with how cholesterol research reshaped cardiovascular medicine over decades will recognize the significance of a single drug moving MACE outcomes by that margin.
Then came tirzepatide, from Eli Lilly. It agonizes two receptors simultaneously: GLP-1 and GIP, the other major incretin hormone. In the SURMOUNT-1 trial, participants on the highest dose lost an average of 22.4% of their body weight at 72 weeks — numbers that approach what bariatric surgery achieves.17 Why the dual agonist outperforms the single one is still being resolved.18
The Part Nobody Likes Talking About
Stop taking semaglutide, and the weight returns.
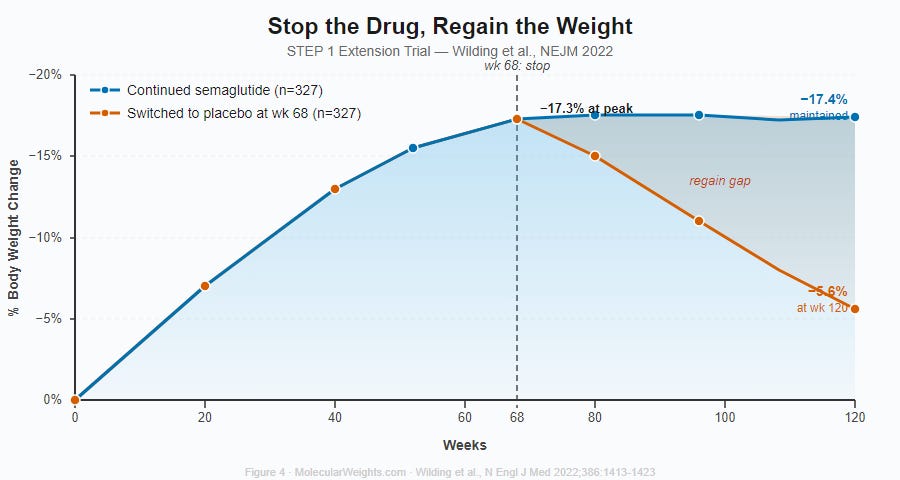
This is not a failure of willpower. The biology predicts it. In the STEP 1 extension study, participants who stopped semaglutide after 68 weeks regained roughly two-thirds of their lost weight within a year.19 In the STEP 4 withdrawal trial, patients who stopped at week 20 gained back nearly 7% of their body weight over the next 48 weeks, while those who continued the drug kept losing.20
Figure 4. Stop the Drug, Regain the Weight. STEP 1 extension trial (Wilding et al., NEJM 2022). Participants who stopped semaglutide at week 68 (orange) regained most of their lost weight within a year — net loss shrank from −17.3% to −5.6% by week 120. Those who continued (blue) maintained −17.4% through week 120. Shaded region shows the widening gap.
What GLP-1 receptor agonists do, in plain terms, is supply pharmacologically the satiety signal the body was not producing effectively enough on its own. When the drug stops, the signal stops. A 2024 paper in Science used optogenetics and human fMRI to identify GLP-1 receptor neurons in the dorsomedial hypothalamus as the substrate for the preingestive satiation these drugs produce — the sense of fullness before you finish eating.21 When you stop the drug, those neurons go quiet, and the old appetite returns.
These are chronic medications. That framing matters enormously for insurance coverage, for public health budgets, and for whether the people who need them most will ever actually have access to them.
💡 Did You Know?
The Gila monster eats only 3–4 times a year. Exendin-4 likely evolved to slow digestion after one of those rare, enormous meals — which is why it mimics a satiety hormone so precisely. The lizard was solving the same problem pharmacologists spent decades trying to crack.
DPP-4 destroys roughly half of all GLP-1 before it ever reaches the bloodstream. The enzyme doesn’t wait for the hormone to circulate — it cleaves GLP-1 in the gut wall and portal vein on the way to the heart. The 2-minute half-life cited in the body of this article is what survives that first pass.
Semaglutide is just three chemical changes away from the native GLP-1 molecule. One amino acid swap at position 8 blocks DPP-4 cleavage. One arginine swap redirects fatty-chain attachment. One C18 fatty diacid chain anchors the whole thing to albumin. That’s the difference between a 2-minute molecule and a 7-day drug.
An oral version of semaglutide already exists. Rybelsus (approved 2019) pairs semaglutide with SNAC — a carrier molecule that briefly permeabilizes the stomach lining to let the peptide through. Getting a protein drug through the stomach without being digested was considered essentially impossible for most of the 20th century.
GLP-1 receptors sit in the heart muscle itself. The 20% reduction in cardiovascular events seen in the SELECT trial may not be entirely explained by weight loss. Direct anti-inflammatory effects on cardiac tissue are under active investigation — and may turn out to be part of why these drugs work so well.
The incretin effect accounts for 50–70% of your entire insulin response to a meal. In type 2 diabetes, that gut-hormone-to-pancreas signal is largely broken. GLP-1 receptor agonists don’t fix the broken signaling — they bypass it entirely by flooding the receptor pharmacologically.
An Unexpected Signal
GLP-1 receptors are not only in the pancreas and gut. They are distributed through the hypothalamus, the brainstem, and the nucleus accumbens — a region central to reward and craving.22 Animal studies have shown GLP-1 agonists reduce consumption of alcohol, cocaine, amphetamine, and nicotine in rodents.23 Early clinical trials suggest similar signals in humans: a 2024 systematic review of five randomized trials found three showing significant reductions in alcohol and nicotine use with GLP-1 agonists.24
The picture is preliminary. As addiction researcher Lorenzo Leggio summarized in Nature Medicine in 2023: “promising but unproven.”25 Semaglutide-specific randomized trials for alcohol use disorder are underway but have not yet reported their primary outcomes. This may be the next chapter of the story, or it may not hold up at scale.
Either way, the trajectory since 1992 has a shape worth noting. John Eng ran a chemical screen through the venom of a Sonoran Desert lizard, found a molecule he described precisely but couldn’t explain, and his careful final sentence — predicting the mammalian analog — pointed at a hormone whose therapeutic consequences we are still mapping thirty years on.
The biology was there all along. It took a lizard to show it to us.
References
Eng J et al. (1992). Isolation and characterization of exendin-4, an exendin-3 analogue, from Heloderma suspectum venom. J Biol Chem 267(11):7402–7405. PMID 1313797
Göke R et al. (1993). Exendin-4 is a high potency agonist and truncated exendin-(9-39)-amide an antagonist at the glucagon-like peptide 1-(7-36)-amide receptor of insulin-secreting beta-cells. J Biol Chem 268(26):19650–19655. PMID 8396143
Tian L et al. (2016). The incretin hormone GLP-1 and mechanisms underlying its secretion. J Diabetes 8(6):753–775. doi:10.1111/1753-0407.12398
Lee YS & Jun HS (2014). Anti-diabetic actions of glucagon-like peptide-1 on pancreatic beta-cells. Metabolism 63(1):9–19. doi:10.1016/j.metabol.2013.08.003
Nauck MA et al. (2018). Incretin hormones: Their role in health and disease. Diabetes Obes Metab 20(Suppl 1):5–21. PMID 29364588
Nauck MA et al. (2019). GLP-1 receptor agonists in the treatment of type 2 diabetes — state-of-the-art. Eur J Endocrinol 181(3):R211–R234. doi:10.1530/EJE-19-0167
Eng J et al. (1992) — see ref. 1. Exendin-4 DPP-4 resistance due to Gly at position 2; half-life ~2.4 h confirmed in pharmacological studies.
Göke R et al. (1993) — see ref. 2.
Nauck MA et al. (2020). GLP-1 receptor agonists in the treatment of type 2 diabetes: state-of-the-art. Mol Metab 46:101102. doi:10.1016/j.molmet.2020.101102
Nauck MA et al. (2020) — see ref. 9.
Knudsen LB & Lau J (2019). The Discovery and Development of Liraglutide and Semaglutide. Front Endocrinol 10:155. doi:10.3389/fendo.2019.00155
Lau J et al. (2015). Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide. J Med Chem 58(18):7370–7380. PMID 26308095
Hall SF et al. (2018). Clinical Pharmacokinetics and Pharmacodynamics of Semaglutide. Clin Pharmacokinet 57(10):1313–1328. doi:10.1007/s40262-017-0618-7
Wilding JHH et al. (2021). Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP 1). N Engl J Med 384:989–1002. PMID 33567185
Garvey WT et al. (2022). Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nat Med 28(10):2083–2091. PMID 36216945
Lincoff AM et al. (2023). Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes (SELECT). N Engl J Med 389:2221–2232. PMID 37952131
Jastreboff AM et al. (2022). Tirzepatide Once Weekly for the Treatment of Obesity (SURMOUNT-1). N Engl J Med 387:205–216. PMID 35658024
Nauck MA et al. (2021). The evolving story of incretins (GIP and GLP-1) in metabolic and cardiovascular disease. Diabetes Obes Metab 23(Suppl 3):5–29. PMID 34310013
Wilding JHH et al. (2022). Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes Obes Metab 24(8):1553–1564. PMID 35441470
Rubino D et al. (2021). Effect of Continued Weekly Subcutaneous Semaglutide vs Placebo on Weight Loss Maintenance (STEP 4). JAMA 325(14):1414–1425. PMID 33755728
Park JS et al. (2024). GLP-1 increases preingestive satiation via hypothalamic circuits in mice and humans. Science 385(6707):438–446. PMID 38935778
Alhadeff AL et al. (2012). GLP-1 neurons in the nucleus of the solitary tract project directly to the ventral tegmental area and nucleus accumbens to control for food intake. Endocrinology 153(2):647–658. doi:10.1210/en.2011-1808; Dossat AM et al. (2011). Glucagon-Like Peptide 1 Receptors in Nucleus Accumbens Affect Food Intake. J Neurosci 31(42):14915–14922. doi:10.1523/JNEUROSCI.3262-11.2011
Volkow ND & Xu R (2025). GLP-1R agonist medications for addiction treatment. Addiction 120(2):198–200. PMID 39049203
Martinelli S et al. (2024). Potential role of glucagon-like peptide-1 (GLP-1) receptor agonists in substance use disorder: A systematic review of randomized trials. Drug Alcohol Depend 264:112424. PMID 39288591
Leggio L et al. (2023). GLP-1 receptor agonists are promising but unproven treatments for alcohol and substance use disorders. Nat Med 29(12):2993–2995. PMID 38001271