Sin Nombre: The Virus Without a Name
In the spring of 1993, a mystery illness killed two healthy young Navajo people in New Mexico and stumped the CDC. Three decades later, its closest relative is on a quarantined ship in the Atlantic.
Sin Nombre: The Virus Without a Name
In the spring of 1993, a mystery illness killed two healthy young Navajo people in New Mexico and stumped the CDC. Three decades later, its closest relative is aboard a quarantined ship in the Atlantic.
On the morning of May 11, 1993, a 19-year-old Navajo man who competed as a runner arrived at a small hospital in Crownpoint, New Mexico. He had a fever. His muscles ached. Within hours, fluid was filling his lungs. By the time doctors understood what they were dealing with, he was dead from respiratory failure.
He should not have died this quickly. Young, fit, no history of illness. When Richard Malone, a deputy medical investigator in New Mexico, began asking questions, he learned something that made the case stranger: the young man’s fiancée had died at the same hospital, five days earlier, with identical symptoms.
Both were young and healthy. They died a week apart with the same presentation.
Malone called the CDC.
Disease Without a Name
What followed was one of the more remarkable disease investigations of the twentieth century. The investigation drew on molecular biology and field epidemiology, but the most important ecological intelligence came from Navajo medicine men who had been tracking the pattern for generations. It ended with a virus named for the fact that no one could agree on what to call it.
Thirty-three years later, that same virus’s closest relative is making headlines from the deck of a quarantined ship in the Atlantic.
Patricia McFeeley, a pathologist at the University of New Mexico, performed autopsies on both patients. Severe pulmonary edema: lungs saturated with fluid. No clear cause. Not bacterial pneumonia. Not influenza. The immune response looked wrong in a way she hadn’t seen before.
Bruce Tempest, the medical director at Gallup Indian Medical Center, heard about McFeeley’s findings and started pulling records. He’d treated similar cases before. A pattern, across years, in the same region.
The CDC team that arrived included Jay Butler as overall lead, Robert Breiman directing fieldwork, and C. J. Peters running lab analysis. They weren’t coming to a recognized outbreak. They came to a mystery: a cluster of deaths from a respiratory illness that didn’t exist in any textbook.
What the Medicine Men Knew
By the time the epidemiologists began interviewing Navajo families, fifty medicine men had already convened their own consultation.
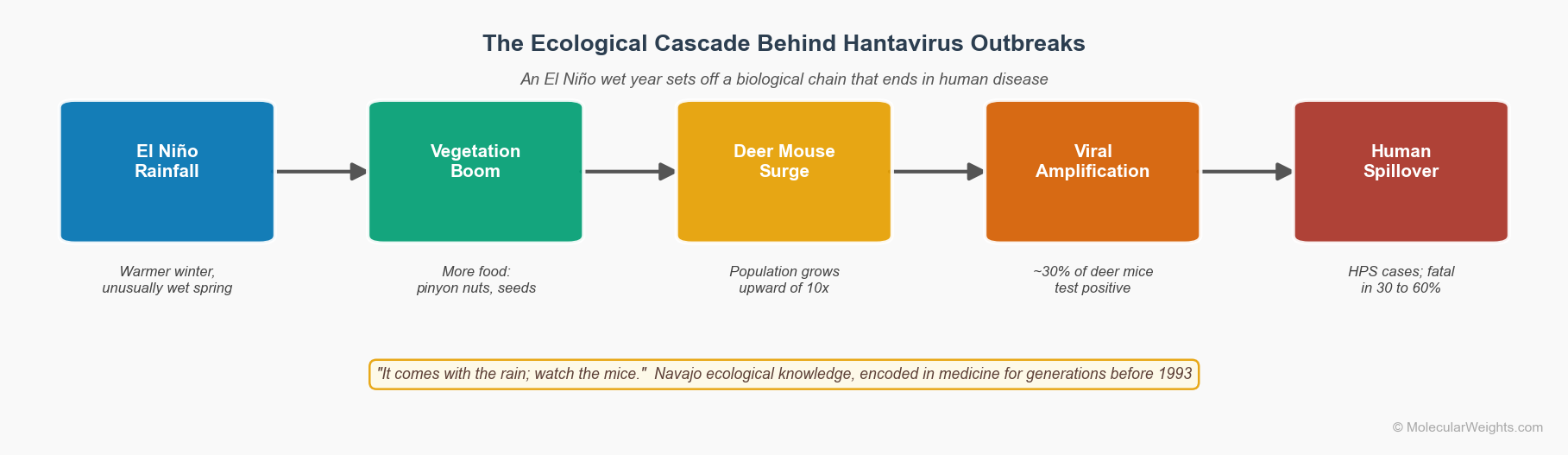
Their message was specific: stay away from deer mice and burn anything contaminated by their droppings. They described similar illness clusters in 1918, 1933, and 1934, each in a wet year, each following a boom in rodent populations. For the elders, the pattern was familiar. A disease that arrived with the rain and retreated with the drought.
The CDC researchers were receiving ecological intelligence the Navajo community had been tracking for a century. Western epidemiology was catching up.
The Mouse and the Weather
In the desert Southwest, dry is the baseline. The El Niño event of 1991–1992 disrupted that. A warmer winter and an unusually wet spring transformed the Four Corners region. Woodlands and scrublands produced more food than usual.
Deer mice noticed.
A University of New Mexico research team documented the result: the western deer mouse population grew significantly between May 1992 and May 1993. Higher rodent density meant the virus spread more easily through the mouse population. More mice also meant more contact with people, more droppings in attics and outbuildings.
Hantavirus doesn’t travel through bites or direct touch. It moves in aerosolized particles from rodent urine and dried feces. Walk into a confined space where an infected mouse has been, disturb the dust, breathe it in. The exposure takes seconds and leaves no trace.
CDC teams spent that summer trapping roughly 1,700 rodents across the affected area. About 30% of western deer mice tested positive for the virus.[1]
Figure 1. The ecological cascade linking El Niño rainfall to human hantavirus cases in the American Southwest. Navajo medicine men described this same chain a century before epidemiologists modeled it.
Identifying the Culprit
On June 10, researchers used reverse transcription polymerase chain reaction to pull viral genetic sequences from tissue samples of the deceased. Six days later, they matched those same sequences in the trapped deer mice.
What they had was a hantavirus. But not one anyone had described before.
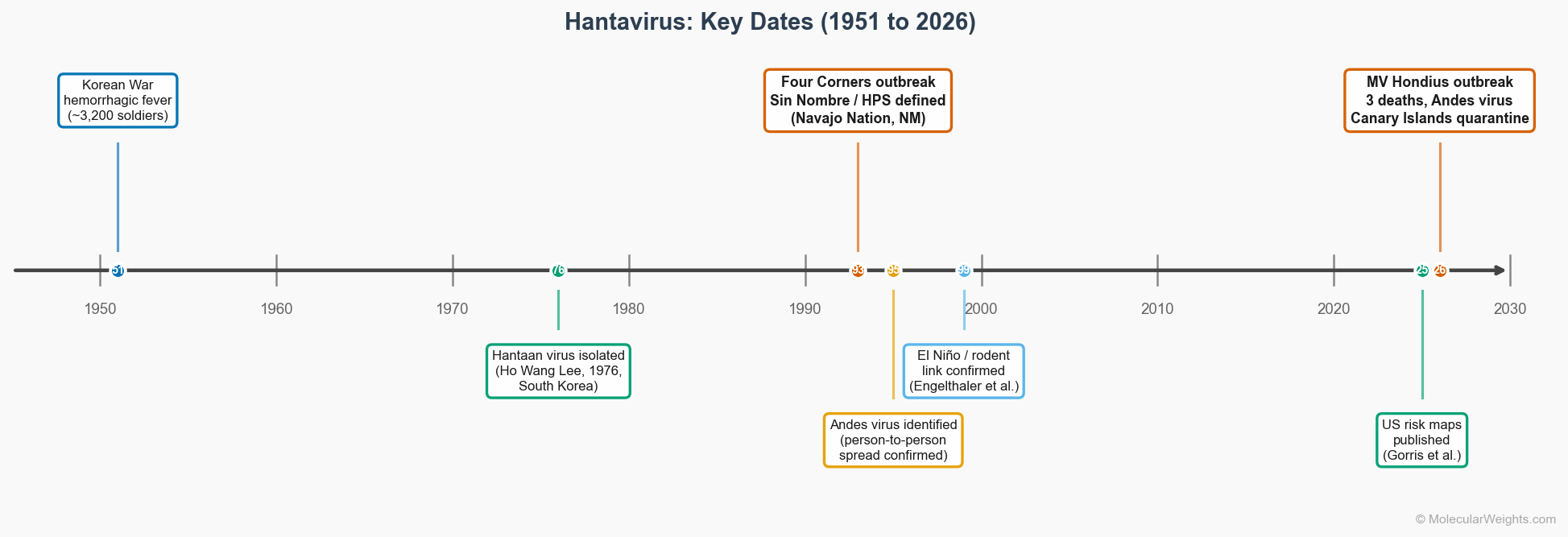
The known hantaviruses came from Asia and Europe. Hantaan virus had been isolated in Korea in 1976, named after the Hantan River where UN soldiers contracted a mysterious hemorrhagic fever in 1951 (roughly 3,200 soldiers fell ill during the Korean War). Ho Wang Lee spent years proving the cause, finally demonstrating in 1976 that lung antigens from Korean field mice reacted with antibodies from war survivors. The virus was formally named in 1980. The kidney-destruction syndrome it caused (hemorrhagic fever with renal syndrome, or HFRS) had been killing soldiers and farmers for centuries; Western medicine had simply lacked a name for the pathogen.
The Four Corners variant was different. Instead of attacking the kidneys, it attacked the lungs. Hantavirus Pulmonary Syndrome had not existed as a clinical entity until that summer. In the most literal sense, it was a new disease.
Figure 2. Key dates in hantavirus research, 1951 to 2026. The 1993 Four Corners outbreak and the 2026 MV Hondius incident (both red) bracket three decades of discovery.
Sin Nombre
Naming it became its own problem.
“Four Corners virus” was proposed first. Residents of four states objected. The CDC tried “Muerto Canyon virus,” chosen because the canyon near the outbreak site appeared on existing maps. The Navajo Nation Council voted unanimously against it. The canyon’s name memorialized a Spanish massacre of 115 Navajo people in 1805. Attaching that place name to a newly discovered disease was not a neutral act.
The naming committee reconvened. “Convict Creek virus” was proposed and failed for technical reasons. The researchers were out of options.
They settled on Sin Nombre virus, Spanish for “without a name.” A pathogen so contentious to label that the scientists named it after the naming problem itself.
What the Virus Does
Sin Nombre doesn’t destroy tissue directly. The damage is immunological.
The viral spike glycoproteins bind to β3 integrin receptors on endothelial cells lining the pulmonary capillaries. Infected cells respond by producing vascular endothelial growth factor (VEGF), which signals capillary walls to increase permeability. Fluid migrates into alveolar spaces.
The immune system arrives. Cytotoxic T lymphocytes release interferon and TNF-α, amplifying the inflammation. The permeability cascade accelerates.
The patient drowns in their own immune response. The virus is the trigger; the fatality follows from the fire.
No antiviral drug has been approved for hantavirus infection. No vaccine exists. Treatment means oxygen, mechanical ventilation, and careful fluid management, and for roughly 30 to 60% of patients who reach the cardiopulmonary phase, that still isn’t enough.[2]
A Global Genus
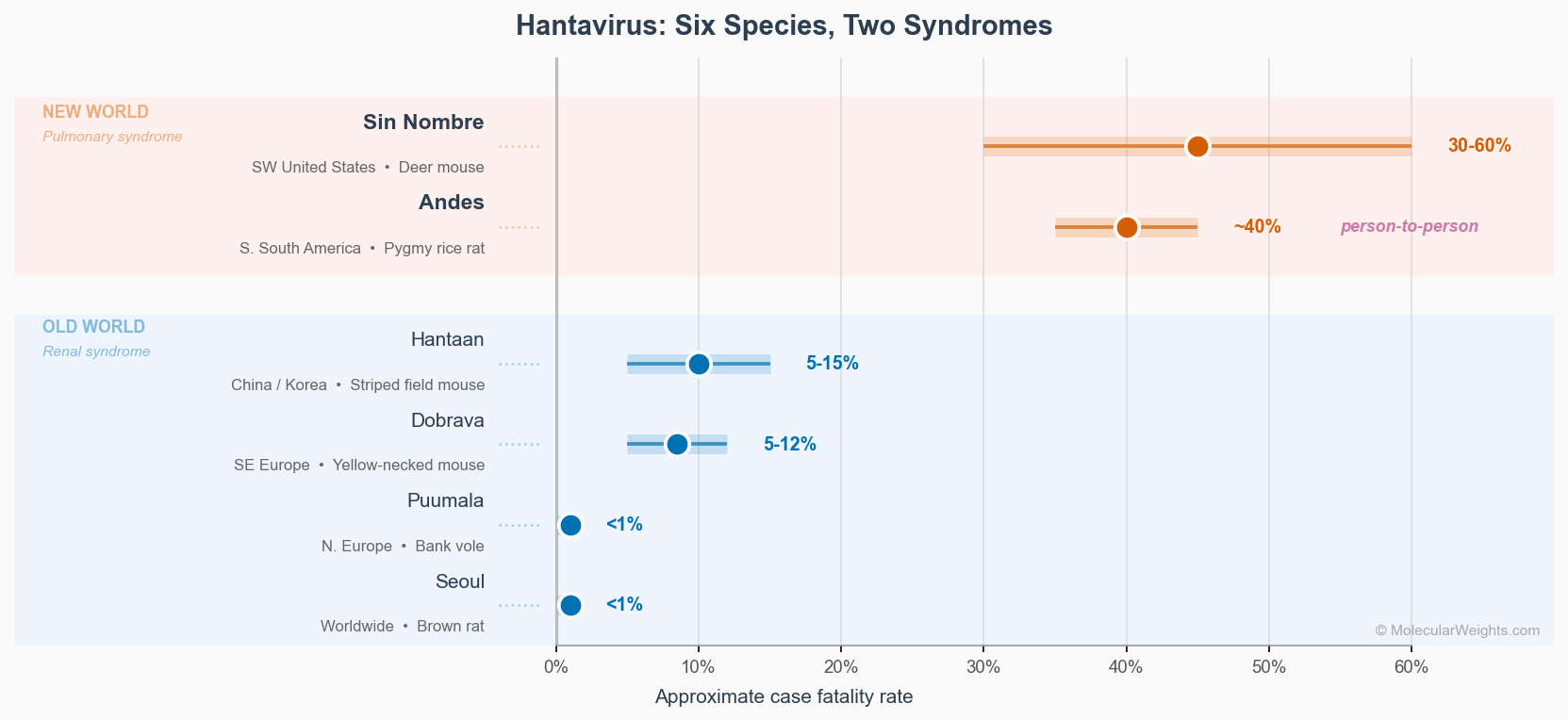
Orthohantavirus now contains 37 classified species, each paired with a specific rodent host. Old World strains circulate across Asia and Europe, causing HFRS rather than pulmonary disease. Puumala, carried by bank voles in Scandinavia and Finland, produces a mild kidney syndrome with less than 1% mortality; Seoul virus travels with brown rats and appears worldwide, including in US laboratory animal facilities. Hantaan and Dobrava-Belgrade are the severe Old World strains, killing 5 to 15% of those they infect in parts of Asia and the Balkans.
Together, Old World strains produce more than 100,000 confirmed HFRS cases per year. That figure is almost certainly an undercount.
The New World strains cause only a few hundred confirmed cases annually, but at far higher case fatality. And one of them carries a property the others lack.
Figure 3. Six hantavirus species ranked by approximate case fatality rate. New World strains (top) cause pulmonary syndrome; Old World strains (bottom) cause hemorrhagic fever with renal syndrome. Andes is the only species with confirmed person-to-person transmission.
Andes: The One That Jumps Between People
Andes virus circulates among long-tailed pygmy rice rats in southern South America, concentrated in the Chilean and Argentine lake districts. Case fatality runs around 40%.
It is, as far as researchers can confirm, the only hantavirus that transmits person-to-person.[3] Every other species in the genus is a dead-end spill from rodent to human. Andes can spread through close contact with an infected person’s respiratory secretions. That property makes it distinctly dangerous in enclosed spaces.
A Ship in the Southern Ocean
On April 1, 2026, the MV Hondius departed Ushuaia, Argentina (the standard embarkation port for expedition cruises to the Antarctic Peninsula and the Patagonian wilderness). By April 11, a passenger was dead. By April 26, a second person had died in Johannesburg, and a third died aboard on May 2.
Swiss authorities confirmed Andes virus in a patient in Zurich on May 6.
As of May 10, the Hondius is anchored near the Canary Islands. All 146 passengers and crew are undergoing retesting. Evacuated passengers are quarantined across multiple countries, with the expected hold running approximately six weeks. The World Health Organization has assessed the global risk as low and continues active surveillance.
How the virus reached the ship isn’t settled. Cruise passengers in Patagonian wilderness routinely encounter structures where reservoir rodents live: shoreline shelters and remote wilderness huts. All cases may trace to common environmental exposure at the same sites. Given Andes’s documented person-to-person capacity, however, someone infected early may also have been a secondary source aboard.
The investigation is ongoing.
Did You Know?
· Hantaviruses have co-evolved with their rodent hosts for so long that viral phylogeny mirrors rodent phylogeny almost exactly. Researchers can sometimes predict which rodent family carries a newly discovered strain from the virus’s genome alone, before the host is trapped and identified.[4]
· Seoul virus, which causes mild kidney disease in humans, was discovered in the 1980s after an outbreak in laboratory rat colonies in the United States and United Kingdom. Several researchers had been handling infected animals for years without knowing it.[5]
· In Scandinavia, public health authorities track bank vole population cycles and issue seasonal hantavirus advisories ahead of predicted rodent peaks. Vole numbers boom and crash on roughly four-year cycles; infection rates in people follow the same rhythm.
· Clinical trials of ribavirin, effective against some hemorrhagic fevers, showed no survival benefit for hantavirus pulmonary syndrome. It helps for HFRS, the Old World kidney-attack form, but not for the lung disease that kills in the Americas.[6]
The Pattern Has Not Stopped
The 1993 outbreak was not a fluke. A 2025 study analyzed 431 US HPS cases from 1993 to 2022 and found that arid conditions with sparse human development and diverse rodent fauna best predicted risk.[7] Four hundred of those 431 cases fell in the West.
When El Niño rains boost vegetation, deer mouse populations surge. Human exposure follows.[8] The drought-to-flood oscillation will happen again. It may already be underway somewhere in the American Southwest.
Treatment in 2026 is not fundamentally different from what Butler’s team recommended in 1993. Oxygen and mechanical ventilation when the lungs fail. That is still the protocol.
What has changed is our understanding of where the virus lives and what drives it into contact with people. The 2025 risk maps give public health agencies the ability to anticipate the next outbreak location rather than just respond to it. The Navajo medicine men had the same insight a century before the modeling: it comes with the rain; watch the mice.
Sin Nombre. The virus without a name. It was living in the deer mice of the American Southwest long before anyone gave it a disease category. The name records the argument over what to call it. The virus, meanwhile, was never confused about what it was.
[1] Jacob AT et al. (2023). Sin Nombre Virus and the Emergence of Other Hantaviruses: A Review of the Biology, Ecology, and Disease of a Zoonotic Pathogen. Biology (Basel) 12(11):1413. PMID 37998012
[2] Jacob AT et al. (2023). Sin Nombre Virus and the Emergence of Other Hantaviruses: A Review of the Biology, Ecology, and Disease of a Zoonotic Pathogen. Biology (Basel) 12(11):1413. PMID 37998012
[3] CDC. (2024). Hantavirus. Centers for Disease Control and Prevention. cdc.gov/hantavirus (last reviewed May 13, 2024)
[4] Jacob AT et al. (2023). Sin Nombre Virus and the Emergence of Other Hantaviruses: A Review of the Biology, Ecology, and Disease of a Zoonotic Pathogen. Biology (Basel) 12(11):1413. PMID 37998012
[5] Jacob AT et al. (2023). Sin Nombre Virus and the Emergence of Other Hantaviruses: A Review of the Biology, Ecology, and Disease of a Zoonotic Pathogen. Biology (Basel) 12(11):1413. PMID 37998012
[6] CDC. (2024). Hantavirus. Centers for Disease Control and Prevention. cdc.gov/hantavirus (last reviewed May 13, 2024)
[7] Gorris ME et al. (2025). Hantavirus is Associated With Open Developed Areas and Arid Climates, Highlighting Increased Risk in the Western United States. Transbound Emerg Dis 2025. PMID 41141578
[8] Engelthaler DM et al. (1999). Climatic and environmental patterns associated with hantavirus pulmonary syndrome, Four Corners region, United States. Emerg Infect Dis 5(1):87-94. PMID 10081675